Frontal Fibrosing Alopecia
FFA is a recently described form of progressive hair loss presenting as a band of erythematous follicles with developing atrophy at the fronto-temporal hairline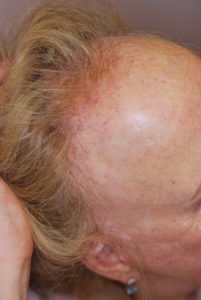
Post menopausal women suffer this irreversible frontal fibrosing alopecia which presents as a marked recession of the frontal-temporal hair line. Pruritis may exist. There is an absence of follicular ostia and mild scarring may present. The scalp may be pale. Eyebrows may be affected.
The cause of this progressive cicatricial alopecia is currently unknown.
A skin biopsy may confirm the diagnosis.
The Trichological Society
Frontal Fibrosing Alopecia – Inga Zemite MD LTTS
1. Introduction
Frontal fibrosing alopecia (FFA) is a primary lymphocytic cicatricial alopecia with a distinctive clinical pattern of hair loss characterized by progressive recession of the frontotemporal hairline, with or without progressive loss of eyebrows.
This particular form of alopecia currently is considered a variant of lichen planopilaris because it is often accompanied by the the presence of cutaneous and/or mucous membrane lichen planus in patients.
FFA is also known as postmenopausal frontal fibrosing alopecia. Female hair loss occurs in more than one pattern.
FFA mainly affects postmenopausal women with a mean age of 67 years.
The exact cause of FFA is unknown. One possible reason can be the disturbed immune response to some component of the scalp hair follicles, however, whether or not the hair loss is caused by hormonal fluctuations is under question.
Histologically, FFA is characterized by a variably dense lymphocytic infiltrate around the infundibulum, isthmus and bulge regions of the affected hair follicles. Inflammation results in loss of sebaceous glands, permanent destruction of the follicle, and its replacement with fibrotic scar tissue. This is manifest clinically as loss of follicular elements.
2. History and Epidemiology
FFA is a quite new clinical entity. It was first described by Kossard in 1994 and that could be regarded as a version of lichen planopilaris with “fronto-temporal” and more rarely, eyebrow hair loss.
Kossard described the first six cases in elderly Australian female patients. Later further cases have been published. This alopecia with characteristic localization and clinical picture was also observed by others. So other cases of FFA have been reported to date, taking into account clinical, histological and pathological features. Till now over 20 cases have been reported in the literature.
Most affected women are postmenopausal, between the age of 40 and 80 years, with a mean age of 67 years. Onset in premenopausal women in their 30s and 40s, and, rarely, in men has also been reported. There are published cases from Australia, Europe, Canada, the Unites States and Korea.
The onset of FFA can occur any time after menopause, whether the menopause is natural or surgically triggered.
3. Pathogenesis
FFA has been classified as a form of LPP based on similar histopathology, and coexistence of classic scalp LPP or lichen planus elsewhere on the body in some subjects.
There are studies that vellus-like and intermediate follicles are more selectively affected, as seen in “fibrosing alopecia in pattern distribution”. While FFA often develops in women with androgenetic alopecia, its onset it also not uncommon at an age well past that accepted for the condition.
Both conditions FFA and androgenetic alopecia often appear to respond to treatment with antiandrogen therapy and topical minoxidil. This suggests a hormonal basis for the disease that may or may not be shared. In FFA the lack of any correlation to peripheral sex hormone levels or the use of hormone supplementation has led to the view that regional factors are involved. These factors appear to be inherent to the aging frontotemporal scalp rather than to hair follicle itself.
4. Clinical features
The disorder presents as a band-like, symmetric anterior alopecia along the frontal and frontotemporal hairline that progressively spreads to the temporal parietal scalp. Symptoms may include mild itch, but generally it is seen rarely. The depth of recession can be from 0,5-8 cm. Loss of follicular ostia can be hard to appreciate. A few remaining hairs may be contained in the recession area. The new hairline is serrated and it often contains hairs with perifollicular erythema and mild hyperkeratosis. The affected skin is atrophic, shiny, and often lighter than the chronically sun-exposed forehead skin, which permits one to quess where the frontal hairline originated.
The pull test is usually negative. Variants include those with midline frontal hairline loss and alopecia that wraps around the entire marginal scalp.
In FFA, the eyebrows are often thinned and may even be absent, it is presenting with lateral or complete eyebrow loss, sometimes with perifolficular and interfollicular erythema.Thinning of axillary, pubic, extremity and abdomen hair, sometimes associated with follicular keratosis and/or erithema, can also be observed.
Classic LPP in other scalp areas and lichen planus elsewhere on the body may coexist. Androgenetic alopecia is a common second diagnosis.
FFA is generally insidious and slow in progression, but rapid loss has also been described in certain cases. Hair loss eventually stops after several years.
5. Laboratory investigations and Pathology
The diagnosis of FFA is usually made on the basis of clinical findings, and laboratory tests are rarely required.
Pathology
Routine evaluations when assessing FFA reveal features that are not easily distinguishable from classic lichen planopilaris and the other lymphocyte-mediated cicatricial alopecias (with the exception of chronic cutaneous lupus erythematosus). Scalp biopsy specimens from the frontal hair margin show perifollicular fibrosis and lymphocytic inflammation concentrated around the isthmus and infundibular areas.
In addition to an important lymphocytic infiltrate around the isthmus and infundibular zone, with ring-pattern perifollicular concentric fibrosis, the external epithelial sheath can show vacuolar degeneration of the basal layer and individual necrosis of the keratinocytes.
In turn, in the inferior portion there are no signs of important inflammation, while the interfollicular epidermis remains intact. The difference with lichen planopilaris is that FFA not only preserves the interfollicular epidermis, but there is also more apoptosis and less inflammation. The infiltrate usually affects the intermediate follicles and vellus hair.
Immuno-phenotyping of the lymphocytes shows a dominance of activated T-helper cells. Direct immunofluorescence, and immunohistochemical studies resemble classic LPP, but, as already said, vellus-like and intermediate hairs appear to be more commonly affected than terminal hairs.
6. Differential diagnosis
FFA must be differentiated from:
- other forms of fibrosing alopecia (DLE, folliculitis decalvans, keloid acne) and lichen pilaris. FFA is distinguishable by a distinctive symmetrical fronto-temporal distribution and a progressive course. Non-scarring, apparently non- inflammatory symmetric hair loss is a characteristic feature of FFA.
- androgenetic alopecia. Frontotemporal androgenetic alopecia lacks the associated inflammatory changes at the hair-bearing edge and is, in general, less band like in pattern. As a rule, androgenic alopecia in female patients does not shift the frontal hairline, but rather affects the vertex and the temples.
- ophiasis. The finding of follicular inflammation at the marginal edge of the alopecia or in the eyebrows is a differentiating feature, seen only in FFA.
- alopecia areata may lead to diagnostic error due to eyebrow hair loss and fold sign positivity (Jacquet’s sign). However, the presence of alopecia plaques in other areas, the lack of cicatricial tissue, the presence of follicular hyperkeratosis and, lastly, the existence of a peribulbar lymphocytic infiltrate serve to establish the difference. It should be taken into account that alopecia areata may be associated with FFA.
- traction alopecias that also lead to progressive miniaturization of the follicles. Differentiation from traction alopecia can usually be suspected from the history, supported by the finding of broken hairs in various lengths in the affected area.
- senile alopecia and chronic telogen effluvium. These conditions do not present the distribution associated with FFA, though the former may show loss at eyebrow level – the latter being progressive and characteristically found in women lacking dense eyebrows earlier in life. In turn, telogen effluvium may be frontovertical, thereby resembling FFA, though temporal alopecia is not seen, and atrophy even less so.
- the familiar high frontal hairline, which is more frequent in women.
The ability of the clinician to recognize that the hair loss process has a scarring or non-scarring nature is critical for accurate diagnosis. The greater is the overlap of clinical and histological features of the conditions, the greater is the diagnostic complexity. If histological features are non-specific, it points to more than one clinical entity is possible. So the differential diagnosis often requires a clinical – pathological confrontation.
7. Treatment
When choosing the treatment it is important to remember that FFA is an irreversible process with a slow course. Currently, there are no evidence-based studies to guide treatment and there is no clearly defined line of treatment for the condition. Therefore treatment options vary among clinicians. Findings of the scalp biopsy, information of the type of inflammation present, location and amount of scalp changes all determine the degree of activity and the selection of appropriate therapy.
Progress of FFA can be arrested by use of moderate potency topical steroids, but it not work always. Treatment should be provided with infiltrations or topical application of corticosteroids. The possible accompanying FAGA in turn should be treated with 5% minoxidil and finasteride at a dosage of 2.5 mg a day.
Recommended first-line therapy is lesional triamcinolone injections {2,5-10 mg/ml every 4-6 weeks or 20mg/ml every 3 month}; this can help to slow the hair loss, although not reliably. Differences in individual response may relate to stage of disease at the time of treatment. Atrophy may be caused by this approach. If there is not response obtained a short course of oral prednisone (0,5-1mg/kg per day for 1-3 month) or chloroquine (150mg/day for 3-9 month) may be tried.
Recently two groups showed that combination oral finasteride (2,5 mg/day) and topical minoxidil (2%-5% twice daily) can arrest hair loss in some women, after 1-2 years of use. So in addition of lesional triamcinolone injections it can further improve response.
In cases where the condition is rapidly progressive, oral prednisone or chloroquine may temporarily slow down the advancement of the disease.
Hormone replacement therapy does not alter the rate of the progress of the disease.
In 2010 Chiang et al. introduced the LPPAI to analyse pre- and post-treatment response with hydroxychloroquine in 40 patients with LPP, including 11 with FFA. The data showed a statistically significant reduction in LPPAI scores at 6 months with continued significant reduction in LPPAI scores at 12 months. Data indicate that hydroxychloroquine significantly reduces signs and symptoms in patients with FFA after both 6 and 12 months of treatment. In FFA the maximal benefits of hydroxychloroquine are seen within the first 6 months of therapy.
There is study described that is done at the UCSF Hair Center.
Systemic treatment with hydroxychloroquine or mycophenolate mofetil was initiated in patients with FFA when biopsy specimens show a moderate to dense inflammatory infiltrate. Doxycycline was considered when the infiltrate was sparse.
Of the 25 patients finallly included in that study, fifteen (64%) were with hydroxychloroquine, four (16%) with doxycycline, and five (20%) with mycophenolate mofetil.
Hydroxychloroquine
Eleven of the 15 patients (73%) showed reduction in signs and symptoms at 6-month follow up. Of these, four (36%) were considered responders and seven (64%) were partial responders. Four of the 15 (27%) showed no response. At 12-month follow up, eight of the nine patients (89%) had reduction in signs and symptoms. Of these, five (62%) were responders and three (38%) were partial responders. One of nine patients (11%) had not responded to treatment at 12-month follow up.
Doxycycline
The four patients of doxycycline were treated for a median of 18 months. At 6-month follow up, two (50%) had a reduction in signs and symptoms. One was a responder and one was a partial responder. Two of four (50%) were nonresponders. Only three patients returned for 12-month follow up. One of the three was a responder, one was a partial responder, and one remained a nonresponder.
Mycophenolate Mofetil
Five patients were treated with mycophenolate mofetil for a median of 6 months. At 6-month follow up, three (60%) patients had responded to treatment. Of these, one (33%) was a responder and two (67%) were partial responders.
None of the patients on hydroxychloroquine experienced any adverse event, including no retinopathy. Three patients experienced an adverse event with the use of doxycycline. Two of these patients had a photosensitive reaction, while one experienced gastrointestinal symptoms. None of the patients reported an adverse reaction to mycophenolate mofetil.
In conclusion must be emphasized that Hydroxychloroquine is significantly effective in reducing signs and symptoms in patients with FFA and has its maximal benefit within the first 6 months of treatment.
8.References
- Blume-Peytavi, U. at all. Hair Growth and Disorders. Springer-Verlag Berlin Heidelberg. 2008.
- Burns, T. at all. Rook’s Textbook of Dermatology. Blackwell Publishing Ltd. 2010.
Frontal Fibrosing Alopecia
Dr Pedro Madureira LTTS
Introduction
Frontal fibrosing alopecia (FFA) is a primary lymphocytic cicatricial alopecia first described by Kossard in 1994. Since then, extensive research had emerged on this specific type of alopecia, but its true prevalence, pathogenesis, associated clinical findings, prognosis and treatment remain unclear. It was regarded as a postmenopausal female hair disease in the spectrum of lichen planopilaris (LPP) afflicting the frontotemporal and/or frontoparietal region with a symmetric and progressive hairline recession. In fact, and although more common in postmenopausal women, reports of FFA in younger women and even in men are increasing. The loss of the eyebrows was recognized as a common and helpful diagnostic feature, often a presenting isolated sign. There is currently no cure for FFA; however, treatment could stop or slow hair loss in some cases.
- Aetiology
The aetiopathogenesis of FFA remains unknown, although hormonal factors, autoimmunity, genetic susceptibility, and some exogen factors are thought to play a role.
The loss of the immune privilege of the hair follicle would be the starting point in the development of scarring alopecias. This bulge immune privilege collapse may be induced by IFN-γ. In FFA, a Th1-biased cytotoxic T cell autoimmune reaction against the hair follicle in the infundibular region and, to a more variable degree, the isthmic region, seems to play a major part. This damage would include the bulge area, where stem cells are placed, leading to a loss of the regenerative potential of the hair follicle and its destruction. A decrease to the absence of labelling with Ki-67, a proliferative marker, and a downregulation of the hair follicle epithelial progenitor cell marker keratin 15, within the bulge area, have already been described in LPP. The melanocyte of the hair follicle might be an antigenic target in FFA; this is supported by the lower melanocyte count found in the upper follicle in lesional skin from FFA patients (not seen in LPP).
1.1. Hormones
Hormonal factors are thought to play a role in FFA, due to their higher frequency in women, especially postmenopausal ones, and their response to 5-alpha reductase inhibitors. Oestrogens produce a decrease in the hair shaft growth and favour the catagen to telogen transition. Therefore, the decrease of oestrogens due to physiological or surgical menopause could alter the control of the hair cycle and be the trigger for the inflammatory attack on the hair follicle in susceptible patients.
Early menopause could be an issue involved in the premature development of FFA or may imply a higher risk of developing FFA, however the course of the disease appears to be unaltered when hormone replacement therapy is introduced. The use of an intrauterine device as a contraceptive may protect against the development of FFA, whereas the intake of tamoxifen with the induction of a low-estrogen environment around the hair follicle may trigger or maintain the pathogenic process of FFA. A few male patients have been reported as having FFA and a history of prostate cancer treated with neoadjuvant hormonal therapy (antiandrogens or estrogens) before the onset of FFA.
A recent study about hormonal dysfunction found that LPP is associated with androgen excess (testosterone or DHEAS), whereas FFA is related to androgen deficiency. According to these findings, DHEAS and androstenedione have been found to be lower in women with FFA compared to a control group. It is possible that the benefits obtained by the treatment of FFA with 5a-reductase inhibitors are the result of DHEA impediment in reaching its final conversion to dihydrotestosterone.
1.2. Associated Diseases and Autoimmunity
FFA patients, mostly women, have an associated immune disorder; the most frequent are thyroid diseases, especially hypothyroidism (8–44.6%). Several studies have demonstrated that patients with FFA are significantly more likely to have systemic lupus erythematosus. Vitiligo and FFA are sometimes associated, and both diseases may share common pathogenic pathways.
A high prevalence of atopy (43.9%) has been demonstrated in a FFA patients cohort. However, according to different studies, one of the most frequently associated cutaneous condition seems to be rosacea, with a prevalence of 15 to 61%.
Androgenetic alopecia (AGA) is observed concomitantly with FFA in 16 to 57% of women and in 67 to 83% of men.
| Autoimmune Associated Diseases |
| Thyroid disorders: Hypothyroidism (8–44.6%)
Hashimoto thyroiditis (8.1%) Graves disease (1.4%) Lichen planus (1.7–18.2%): Cutaneous (3–6.5%) Mucosal (3–16.7%) Pilaris (0.8–25.3%) Psoriasis (7.4%) Vitiligo (0.6–5.6%) Inflammatory bowel disease (5.4%) Lichen sclerosus (0.3–5.4%) Sjögren syndrome (1.7–4.1%) |
1.3. Genetic Factors
Since the first report of familial FFA in 2010, more cases have been published, the largest series so far being one including 20 cases from nine different families. A Human leukocyte antigen (HLA)-DR1 has been related. A study including 13 cases of familial FFA found that most of the patients of that cohort shared HLA-A*33:01; B*14:02; C*08:02, suggesting that this haplotype may predispose to familial FFA.
A recent GWAS demonstrated a significant association with FFA in four genomic loci: 2p22.2, 6p21.1, 8q24.44, and 15q2.1. Fine mapping within the 2p22.2 and 6p21.1 loci revealed associations with a presumed casual missense variant in CYP1B1 (which encodes a member of the cytochrome P450 family involved in the oxidative metabolism of oestrogens) and the HLA-B*07:02 allele, respectively.
The occurrence of the disease in families can indicate exposure to a common environmental trigger, probably enhanced by a genetic predisposition. An autosomal dominant transmission with reduced penetrance has been proposed as a hypothetic inheritance pattern in FFA. With regards to a family history of the disease, this is reported by 5 to 8% of patients with FFA.
1.4. Surgical Procedures and Hair and Skin Care Products
Some cases of FFA or LPP developed after hair transplantation for AGA, so hair transplants may also be affected. Explanations for these situations include the Koebner phenomenon induced by surgical trauma, or an autoimmune attack from a follicle antigen liberated during surgery or induced by a post-surgery pro-inflammatory environment.
The use of sunscreens has been proposed as a possible trigger for the development of FFA since the publication of a study that found a higher use of these products in FFA patients compared to a control group. Concordant results across multiple population-based studies suggest that a true correlation between sunscreen use and FFA may exist, but this does not necessarily imply causation. The higher usage may simply reflect a new behaviour adopted because of the alopecia.
The frequency of shampooing has been found to be significantly lower in patients with FFA compared to a control group; this may reflect the common concern that hair washing may worsen hair loss or suggest the possibility that frequent shampooing reduces the risk of developing FFA by achieving a more efficient removal of exogenous particles that could penetrate the follicular infundibulum and trigger the inflammatory response in patients who are genetically predisposed.
1.5 Other
Regarding dietary habits, a greater consumption of buckwheat and millet groats has been reported in FFA patients, although subsequent studies have not found any association between phytoestrogens or natural PPAR-γ agonists (i.e., grapes) and FFA.
A relationship between occupational exposure to alkylphenolic compounds in women with FFA has been described; these substances have been shown to interact with PPAR-γ and inhibit transformation of DHEA to DHEAS.
The occurrence of an intense and stressful event just before the onset of FFA has been referred to by 76% patients.
- Clinical Presentation
FFA is characterized by frontal and temporoparietal hairline recession, leading to a cicatricial band, which tends to contrast with the photo-aged skin of the superior forehead. The occipital area may also be involved (15–30.4%). In men, the loss of sideburns may be the only sign of the disease. The alopecic area appears as a shiny, atrophic, and pale band of incomplete hair loss. When the original hairline is missing, the manoeuvre of cocking both eyebrows may help to find it: a sharp muscular demarcation is noted between the forehead and the scalp. Wood’s light examination may also help to define the missing hairline. The hairline recession is usually bilateral and symmetric, but asymmetric forms have also been described. Advanced cases can lead to a “clown alopecic pattern”, with total hair loss in the frontoparietal area. Three clinical patterns of FFA, established according to frontal hairline recession, have been described:
- Pattern I (linear) Uniform band of frontal hairline recession in the absence of loss of hair density behind the hairline
- Pattern II (diffuse) Diffuse or zigzag band-like alopecia affecting the frontal hairline with significant loss of hair density behind the hairline (at least a 50% decrease in normal hair density) with a compatible trichoscopy.
- Pattern III (pseudo-fringe-sign) Unaffected primitive frontal or temporal hairline forming the pseudo “fringe sign.”
Figure 1 (a) Pattern I: linear and uniform hairline recession, without loss of hair density behind the new hairline. (b) Patter II: diffuse alopecia behind the frontal hairline with loss of hair density behind. (c) Pattern III: unaffected primitive frontal hairline followed by an alopecic band, forming the pseudo “fringe sign”.
Figure 2 Cocking the eyebrows to find the missing hairline in frontal fibrosing alopecia
The presence of isolated hairs in the original hairline is a helpful diagnostic clue (lonely hair sign). Partial or total eyebrow alopecia is noted in around 63–83% of patients. Eyelash alopecia can also be noted, and in men the beard can also be affected. Clinically non-inflammatory peripheral hair loss, that is, axillary, pubic, and mainly extremity hair loss, is found in 22–77% of patients with FFA, generally occurring before hair scalp loss. Facial papules due to vellus hair involvement are another common finding, they are distributed over the facial skin and are more visible over the temples, although they may also appear on the cheeks or chin.
Other signs described for FFA are the presence of increased pre-auricular lines and increased sweating of the scalp. Regarding symptoms, some patients have pruritus, which seem to be less frequent in the occipital area compared to the frontal hairline.
-
- Diagnosis
| Diagnostic criteria for FFA | |
| Major Criteria | Minor Criteria |
| 1. Cicatricial alopecia of the frontal, temporal, or frontotemporal scalp, in the absence of follicular keratotic papules on the body
2. Diffuse bilateral eyebrow alopecia. |
1. Perifollicular erythema and/or follicular hyperkeratosis, lonely hair sign.
2. Histopathological features of FFA and LPP. 3. Involvement (hair loss or perifollicular erythema) of additional FFA sites (occipital area, facial hair, sideburns, or body hair). 4. Non-inflammatory facial papules. 5. Preceding or concurrent symptoms (pruritus or pain) at the areas of involvement. |
- Histopathology
FFA is characterized by a lichenoid lymphocytic infiltrate around the upper follicle, i.e., isthmus and infundibulum, including the bulge area, as well as concentric perifollicular lamellar fibrosis. A strong correlation between the severity of the peripilar cast and the degree of lymphocytic infiltration has been identified. It is thought that the destruction of the external root sheath at the level of the isthmus is responsible for irreversible alopecia. The lower part of the follicle usually remains spared. A reduction in the number of hair follicles is a consequent finding. The loss of sebaceous glands is also an early finding in FFA. Regarding the composition of the inflammatory infiltrate in FFA, this is characterized by an increase in the percentage of CD8+ T cells, with a reversal of the typical CD4:CD8 ratio.
Although histological findings in FFA seem to be indistinguishable from LPP, a lower melanocyte count has been demonstrated in the upper follicle in FFA patients compared to LPP, and is associated with the hypopigmentation observed clinically in the alopecic band in FFA.
- Differential Diagnosis
Traction alopecia may resemble FFA. The clinical background, with a history of use of tight hairstyles and the absence of typical trichoscopic signs of FFA, may be useful. Moreover, traction alopecia is not associated with eyebrow hair loss. The ophiasis pattern of Alopecia Areata (AA), which affects the margins of the scalp, may masquerade as FFA. In addition, AA may produce eyebrow alopecia, sometimes as an isolated finding. However, perifollicular erythema and hyperkeratosis are absent in AA, whereas yellow dots, dystrophic and broken hairs, black dots, exclamation mark hairs, tapered hairs, and regrowing hairs are common features.
Other scarring alopecias, such as LPP, discoid lupus erythematosus, and pseudopelade of Brocq, tend to produce multifocal alopecic areas. A familial high frontal hairline should also be discarded. AGA with male pattern may also be considered, especially when the frontal or temporal hairline is receded. However, hair miniaturization (with an increased proportion of thin and vellus hair) is not trichoscopic signs in FFA.
- Treatment and Patient Management
-
- Local Treatment
Topical corticoids are recommended, especially in the early inflammatory stage, but relapse occurs upon their discontinuation. However, a study revealed that patients treated with 0.3% tacrolimus were more likely to stabilize in three months compared to clobetasol. Regarding to intralesional corticosteroids, 20 mg/mL of triamcinolone acetonide used in the hairline (every 3–6 months) may obtain hair regrowth in some patients. Topical minoxidil has not shown clinical improvement in the slowing down of the progression of the alopecia.
-
- Systemic Treatments
Oral prednisone (0.5–1 mg/kg/day, three to eighteen months) has been shown to produce a stoppage of hairline recession in almost 43% patients, but with a relapse when the treatment is stopped. An improvement in symptoms and signs of FFA was found in 73% of patients treated with hydroxychloroquine, though most were partial responses. The maximal benefits were seen within the first six months of therapy.
Oral finasteride (2.5–5 mg/day), which inhibits the isoenzime type II of 5-alpha reductase, displayed that 47% of patients showed improvement (regrowth in the hairline) and 53% showed a stabilization of the alopecia. Favourable outcomes have been described with oral dutasteride, which is about three times as potent as finasteride at inhibiting type II 5-alpha reductase and more than 100 times as effective at inhibiting type I.
Oral minoxidil has been demonstrated to improve the background hair thickness in LPP, especially in patients with diffuse LPP, but further studies regarding its efficacy in FFA are needed.
With regards to biological therapies, patients who were treated with oral tofacitinib, a pan-JAK inhibitor, 10–15 mg/day from two to nineteen months, showed a clinical response in 80% of patients and clinical improvement in both FFA patients.
6.3 Patient Management
FFA is usually insidious, but can be rapidly progressive, and may also remain static for periods of time or become stabilized spontaneously after several years of evolution. Without treatment, the hair loss per year, measured by the distance of recession of the hairline, may range from 0.2 to 2.1 cm. In the early disease, eyebrow regrowth may be achieved with some local treatments. The patient’s age and age at disease onset are both predictors of FFA severity, with higher age and age at onset being related to more severe forms.
Higher body mass index has been found to be associated with severe forms, as well as with the presence of rosacea in patients with FFA. Moreover, patients with more severe FFA seem to be more likely to have rosacea. Regarding clinical patterns, pattern III is associated with the best prognosis and pattern II with the worst, whereas pattern I has an intermediate prognosis.

